
It is by mass the most common element on Earth, forming much of Earth’s outer and inner core. Iron is chemically active and forms two major series of chemical compounds, the bivalent iron (II), or ferrous. Iron rusts in damp air, but not in dry air. It is known to exist in four distinct crystalline forms. Iron is a metal in the first transition series. Iron is a lustrous, ductile, malleable, silver-gray metal (group VIII of the periodic table ). Iron sulfate (FeSO 4) is used to treat the blood disease anemia.Įstimated Crustal Abundance: 5. Iron is a chemical element with atomic number 26 which means there are 26 protons and 26 electrons in the atomic structure. Iron plays a role in the creation of chlorophyll in plants and is an essential part of hemoglobin, the substance that carries oxygen within red blood cells. In addition to helping build the world around us, iron helps keep plants and animals alive. Steel is used to make paper clips, skyscrapers and everything in between. 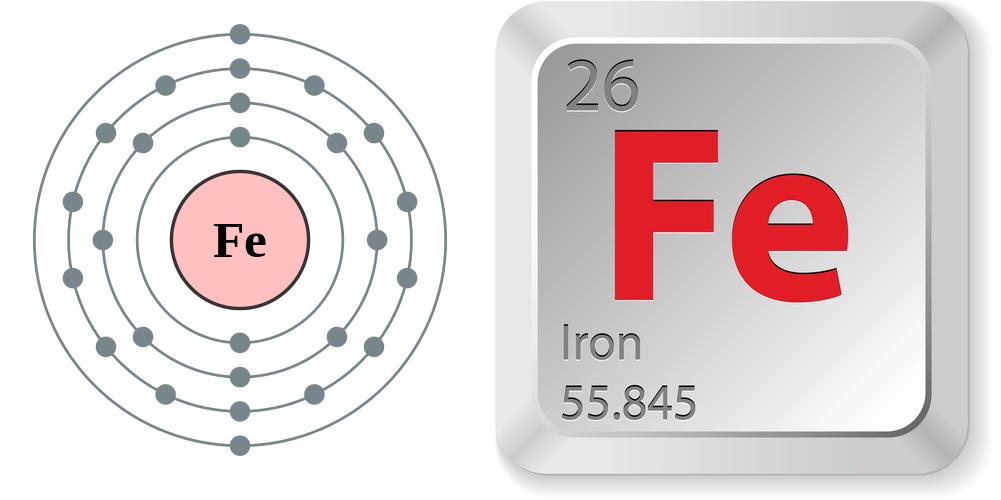
Small amounts of chromium improves durability and prevents rust (stainless steel) nickel increases durability and resistance to heat and acids manganese increases strength and resistance to wear molybdenum increases strength and resistance to heat tungsten retains hardness at high temperatures and vanadium increases strength and springiness. The addition of other elements can give steel other useful properties. Steel typically contains between 0.3% and 1.5% carbon, depending on the desired characteristics. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10-19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. Electrochemical Equivalent: 0.69455g/amp-hr Electron Work Function: 4.7eV Electronegativity: 1.83 (Pauling) 1.64 (Allrod. Iron is a chemical element with atomic number 26 which means there are 26 protons in its nucleus. Huge amounts of iron are used to make steel, an alloy of iron and carbon. Number of Electrons (with no charge): 26 Number of Neutrons (most common/stable nuclide): 30 Number of Protons: 26 Oxidation States: 2,3 Valence Electrons: 3d 6 4s 2 Electron Dot Model. nH 2O) and siderite (FeCO 3) are other important sources.Iron is primarily obtained from the minerals hematite (Fe 2O 3) and magnetite (Fe 3O 4). Iron is the cheapest and one of the most abundant of all metals, comprising nearly 5.6% of the earth's crust and nearly all of the earth's core. An atom of iron has 26 protons in its nucleus therefore, the atomic number of iron is 26 and its ranking on the periodic table of chemical elements is 26. 1 It should not be confused with 'group VIIIA' in the CAS. Group 8' is the modern standard designation for this group, adopted by the IUPAC in 1990. It consists of iron (Fe), ruthenium (Ru), osmium (Os) and hassium (Hs). Therefore, an iron atom has twenty-six protons and twenty. The atomic number of an element is equal to the number of protons and electrons in that element. Iron is the 26th element of the periodic table so its atomic number is 26. Archaeological evidence suggests that people have been using iron for at least 5000 years. Accordingly, the number of protons, which is always equal to the number of electrons in the neutral atom, is also the atomic number. Group 8 is a group (column) of chemical elements in the periodic table. Protons, Neutrons, Electrons for Iron (Fe, Fe2+, Fe3+) Iron is a classified transition metal and its symbol is Fe.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
